I-Mab Provides Business and Corporate Updates and Reports Financial Results for the Six Months Ended June 30, 2022
- Seven key clinical milestones achieved year-to-date, including positive data readouts for lemzoparlimab, uliledlimab, and TJ-CD4B
- Lemzoparlimab is on track for Phase 3 study for 1L MDS
- Amendment to the global partnership with AbbVie for certain new CD47 antibodies currently in development with up to
US$1.295 billion in milestone payments - Significant progress made on the expanded uliledlimab phase 2 NSCLC clinical trial with more complete dataset to be expected
- Continued focus on key value drivers of five clinical-stage assets with three potential BLAs within the next three years
- Repositioned commercialization strategy for felzartamab and eftansomatropin alfa
- Total cash position of
US$586 million (RMB3.9 billion ), sufficient to fund key business operations for more than three years
During this reporting period, the Company has implemented a series of strategic initiatives that aim to re-position its overall business priorities to focus on key value drivers while preserving a strong cash position estimated to be sufficient to support its key business operations for more than three years.
Specifically, the Company will continue to drive value by leveraging its global R&D competitive advantages in immuno-oncology, which has been demonstrated in the Company's short history with such examples as lemzoparlimab, uliledlimab and TJ-CD4B. Today, our pipeline is not only innovative but also advanced as certain key assets move towards BLA and near-term commercialization, including felzartamab and eftansomatropin alfa. The Company's current business model relies on a two-pillar approach to realize the value of innovation. The first pillar is to partner out the global rights of its innovative assets after global phase 1 or phase 2 clinical validation while retaining the rights for clinical development, manufacturing, and commercialization in China. This is exemplified by the global partnership with AbbVie for lemzoparlimab. The Company will continue to pursue more partnership opportunities for other global assets in its pipeline. The second pillar is to rapidly develop the assets towards BLA and commercialization in
Firstly, following a systematic review of its pipeline development and overall business, the Company has prioritized its resources to focus on five key clinical stage assets with 10 ongoing and planned clinical trials. These prioritized programs represent the Company's key value drivers as significant progress in these prioritized programs will not only accelerate the clinical development towards pivotal or registrational studies (such as lemzoparlimab and uliledlimab) but also facilitate global business development deals. The Company is currently pursuing potential global partnership deals for uliledlimab and TJ-CD4B.
Secondly, the Company's current commercialization strategy aims to maximize the commercial value of its pre-BLA products through commercial partnerships typically structured with upfront, regulatory, and sales milestone payments along with significant sales royalties. A commercial partnership for eftansomatropin alfa was established with Jumpcan in 2021 and is progressing as planned. By doing so, the Company is able to avoid investing significant resources to build its sales forces and instead allocate the resources to deliver the value-driver milestones or catalysts. The Company is currently pursuing a similar commercial partnership for felzartamab.
Thirdly, the Company will continue to invest in the discovery and development of its next-generation pipeline assets.
Fourthly, through a strategy of re-positioning the pipeline to prioritize key assets with high probability of commercial success, the Company has been making significant efforts to preserve its strong cash position. As of
"As we weather turbulent market conditions worldwide, we must prioritize our resources to focus on value-driving assets while continuing to deliver on key milestones and strengthening the Company's fundamentals," said Dr.
"On the corporate development front, business development remains a key strategic priority for the Company, and we continue to pursue potential partnership opportunities. Looking ahead, we remain confident in achieving critical clinical and corporate milestones by year-end to continue to create value for our stakeholders."
Updated Pipeline Development Highlights and Upcoming Milestones
The Company's drug pipeline has a number of critical features: (1) The pipeline is innovative and globally competitive, comprised of three generations of products with first-in-class and best-in-class potential. This is exemplified by the first generation of differentiated drug assets, such as felzartamab and eftansomatropin alfa, which are in registrational trial or at a pre-BLA stage, as well as novel monoclonal antibodies such as lemzoparlimab and uliledlimab, which are in phase 2 clinical trials or preparation for phase 3. The second generation of even more innovative bi-specific antibody assets, including TJ-CD4B and TJ-L14B, are in phase 1 clinical trials, followed by additional bi-specific antibody assets progressing towards an IND enabling stage. The new discovery initiatives for the third-generation innovation are on the way for high-risk and high-value drug candidates enabled by transformative technologies. (2) The pipeline is focused on immuno-oncology and biologics, leveraging its unique R&D and CMC strengths. (3) The pipeline is advanced with three assets are either in phase 3 or registrational studies or planned for phase 3. The Company expects to achieve three potential BLA submissions or market launches between 2023 and 2025.
The chart below summarizes the development status of the Company's clinical stage pipeline (pre-clinical programs are not shown).
(1) Five prioritized clinical assets:
Lemzoparlimab (Phase 3 planned): The current focus is on the combination therapy of lemzoparlimab with azacytidine (AZA) for the treatment of newly diagnosed myelodysplastic syndromes (MDS) with the goal of being first-to-market in
Clinical development progress and update on global strategic partnership with AbbVie. Lemzoparlimab, a novel CD47 antibody developed by the Company, is being investigated through a comprehensive clinical development plan for hematologic malignancies and solid tumors. The Company's near-term priority is to initiate a phase 3 registrational trial in newly diagnosed higher-risk MDS patients by Q4 2022, potentially making lemzoparlimab the first CD47 product in
- Lemzoparlimab in combination with AZA for AML and MDS: Over 90 patients with newly diagnosed MDS or acute myeloid leukemia (AML) have been dosed with lemzoparlimab at 30 mg/kg in combination with AZA in
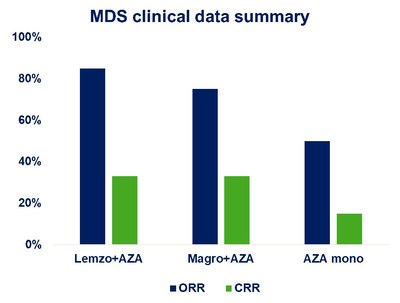
China . This patient cohort had a more severe disease at baseline due to disease conditions and clinical practice patterns inChina .I-Mab's recent data analysis of the MDS cohort, including over 50 patients who received the combination treatment, showed that without a priming dose, lemzoparlimab was well tolerated. The Company observed significant clinical responses as defined by the overall response and complete response rates, which improved over time (Figure 1). Detailed safety and efficacy data, along with gene mutation analysis, will be presented in a proffered paper at theEuropean Society for Medical Oncology (ESMO) Congress 2022.
Phase 3 clinical trial of lemzoparlimab in combination with AZA as a 1L treatment for MDS. An end-of-phase 2 (EOP2) meeting request was submitted to China CDE and NMPA and discussion is ongoing with CDE to initiate a phase 3 clinical trial in patients with MDS by the end of 2022.
Figure 1. Clinical efficacy of lemzoparlimab and AZA combination in MDS patients who received initial dose over six months
Lemzoparlimab in combination with rituximab for non-Hodgkin's lymphoma (NHL): The dose expansion trial is ongoing to enroll more patients. The preliminary data were reported at ASH 2021.
Lemzoparlimab in combination with PD-1 therapy for solid tumors: Phase 2 clinical trial in combination with a PD-1 antibody (pembrolizumab or toripalimab) are ongoing in patients with selected advanced solid tumors.
Update on AbbVie partnership: On
The Company continues its commitment to lemzoparlimab development based on the accumulative safety data, without the need for a priming dosing regimen, from nearly 200 cancer patients as well as the efficacy data, especially from a Phase 2 study of combination therapy of lemzoparlimab and AZA in patients with higher risk MDS. The detailed data will be presented in a proffered paper at the
AbbVie has discontinued the global Phase 1b study of lemzoparlimab combination therapy with AZA and venetoclax, in patients with MDS and AML. This decision was not based on any specific or unexpected safety concerns.
Uliledlimab (End of Phase 2, Pivotal Study Planned in 2023): A highly differentiated CD73 antibody being developed for solid tumor indications. The Company is currently advancing uliledlimab in two phase 2 clinical trials in the
Phase 2 clinical study of uliledlimab in combination with PD-1 antibody (toripalimab) in advanced NSCLC: The Company presented the preliminary clinical results of an ongoing phase 2 clinical study of uliledlimab in combination with toripalimab (TUOYI®) in patients with NSCLC at the 2022
The phase 2 preliminary efficacy data as of
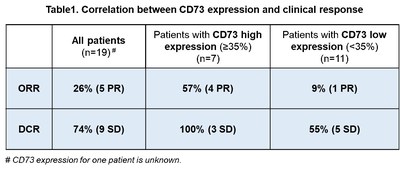
Based on the preliminary data mentioned above, the phase 2 clinical trial was expanded to focus on enrolling the selected patient cohort with advanced NSCLC who were previously ineligible for standard of care treatment for further evaluation of treatment efficacy as well as the role of CD73 as a potential predictive biomarker.
The current status of the expanded phase 2 clinical trial in patients with NSCLC: As of
Development of companion diagnostic (CDx) kit of CD73: Based on the correlation data between clinical response and tumor CD73 expression,
Felzartamab (BLA ready for 3L MM, Phase 3 for 2L MM): A differentiated CD38 antibody for the treatment of relapsing and refractory multiple myeloma (MM) and potentially autoantibody-mediated autoimmune diseases such as membranous nephropathy. A new combination therapy with lemzoparlimab for high-risk MM is being investigated pre-clinically. The Company owns the rights for the development, manufacturing, and commercialization of felzartamab in
Felzartamab is positioned as the first and only locally manufactured CD38 antibody to be commercially more competitive in
China BLA ready for third-line MM: The registrational trial has been completed, and the topline data have met the preset primary and secondary endpoints. More importantly, the clinical data have confirmed the clinical advantages of felzartamab in terms of lower infusion-related reaction rate and shorter infusion time, which has made it possible for its use in an outpatient clinic setting. In
New potential combination therapy with lemzoparlimab: CD47 is consistently and highly expressed on MM tumor cells expressing various levels of CD38; the Company investigated the possibility of combining felzartamab with lemzoparlimab for a more effective treatment for MM in a pre-clinical setting. The results supported that the combination therapy has a synergistic effect on MM. The data have been submitted to ASH 2022 for presentation.
Eftansomatropin alfa (Phase 3, BLA ready in 2023): A differentiated long-acting growth hormone for pediatric growth hormone deficiency (PGHD). Eftansomatropin alfa is the only rhGH in its proprietary fusion protein format (pure protein-based molecule) without chemically linking with PEG or other moieties. Its safety, tolerability, and efficacy have been well demonstrated in a phase 2 clinical trial in the EU. The Company has the rights for the development, manufacturing, and commercialization of eftansomatropin alfa in
Phase 3 clinical trial for PGHD: This phase 3 registrational trial (TALLER) of eftansomatropin alfa as a weekly treatment for PGHD patients is ongoing in
Strategic commercial partnership with Jumpcan: In
TJ-CD4B/ABL111 (Phase 1): A novel Claudin 18.2 and 4-1BB bi-specific antibody is composed of a highly potent Claudin18.2 IgG with high binding affinity even in Claudin18.2 low-expressing tumors and a unique 4-1BB scFv which could stimulate T cells only upon tumor cell engagement to avoid systemic and liver toxicity. TJ-CD4B is designed to treat patients with Claudin18.2 positive gastric and pancreatic cancer. In
Phase 1 clinical trial of TJ-CD4B in patients with advanced or metastatic solid tumors: The dose escalation part of the study reached 8 mg/kg without encountering dose limiting toxicity. More data are being generated as the trial progresses. As of Q2 2022, 5 dose cohorts had been completed, with 16 subjects dosed. Regarding safety, no grade 2 TRAEs or DLTs were reported. There is a dose-dependent increase of drug exposure and soluble 4-1BB in serum, suggestive of a favorable PK/PD profile and potentially a longer dosing interval with durable T cell activation. Preliminary clinical activity was also observed, with one confirmed PR of a metastatic esophageal adenocarcinoma patient who failed three lines of prior therapies, including PD-1 therapy, and three cases of stable disease (SD). The study is currently at 8 mg/kg without significant toxicities. Additional clinical sites in
(2) Other clinical assets
Efineptakin alfa (Phase 2): The world's first and only long-acting recombinant human interleukin-7 ("rhIL-7") and is designed as a monotherapy for the treatment of cancer patients with lymphopenia because of its unique properties of increasing tumor-attacking T cells and as a combination with a PD-1 or PD-L1 antibody because of its potential synergism with PD-1/PD-L1 therapy. The Company has the rights for the development, manufacturing, and commercialization of efineptakin alfa in
Phase 2 Clinical Trial: the first patient was dosed in a phase 2 study of efineptakin alfa (also known as TJ107) in combination with pembrolizumab (Keytruda®) in patients with advanced solid tumors in
Clinical data published by Genexine/
Plonmarlimab (TJM2): a monoclonal antibody targeting human granulocyte-macrophage colony-stimulating factor (GM-CSF), a cytokine that plays a critical role in acute and chronic inflammation and cytokine release syndrome (CRS) associated with CAR-T and severe COVID-19.
CRS associated with severe COVID-19: In
Enoblituzumab (TJ271): A humanized B7-H3 antibody as an immuno-oncology treatment agent. Enoblituzumab works through a dual mechanism to attack tumor cells, i.e., ADCC and immune activation. The Company licensed the rights for the development and commercialization of enoblituzumab in
TJ210/MOR210: A novel monoclonal antibody targeting C5aR1 to treat solid tumors through the suppression of myeloid-derived suppressor cells and modulation of tumor microenvironment in favor of enhanced anti-tumor immune response as a novel mechanism of action. The in vitro and in vivo pre-clinical studies are ongoing to explore and validate the most effective combination partner(s) of TJ210 in addition to the PD-(L)1 antibody.
TJ-L14B/ABL503: A novel PD-L1-based bispecific antibody with the PD-L1 arm to target PD-L1+ tumor cells and block PD-L1/PD-1 interaction and the 4-1BB arm to conditionally activate T cells upon local tumor engagement. Phase 1 clinical trial in patients with advanced solid tumors: Dose escalation of TJ-L14B monotherapy is ongoing in the
(3) Preclinical assets and programs
The Company's R&D strategy to sustain and enrich the growing innovative immuno-oncology pipeline is to generate the next generation of innovative assets enabled by cutting-edge science and technology. The goal is to achieve four to five INDs or phase 1 clinical trials within the next three years by focusing on bi-specific antibodies and so-called "super antibodies" that are enabled by new technologies, such as mRNA technology, or formatted with novel modalities such as the masking technology. Innovative immune adjuvants to activate both innate and adaptive anti-tumor immunity are also being developed. The Company has made steady progress in advancing the development of these preclinical assets culminating in successful candidate selection of two bispecific molecules early this year and another 4 molecules expected to achieve candidate selection in the near future. Two leading molecules are described below.
TJ-L1IF is a novel PD-L1/IFN-α antibody-cytokine fusion protein, which is specifically designed for the treatment of PD-1/PD-L1 resistant tumors through the addition of a strong immune adjuvant (interferon-alpha, IFN-α) to potentially convert "cold" tumor to "hot" tumor on top of a PD-L1 antibody to achieve superior anti-tumor activity than PD-(L)1 antibody monotherapy. Novel drug molecules with such design is badly needed to address the current clinical challenges where a majority of cancer patients do not or poorly respond to PD-1/PDL-1 therapies.
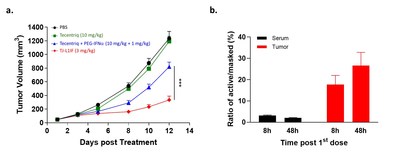
IFN-α was the first cytokine approved for cancer treatment, but its clinical use is highly limited due to considerable systemic toxicity. TJ-L1IF is composed of a PD-L1 VHH nanobody linked with the Fc of human IgG with an engineered IFN-α2b fused at the C-terminus. It is a prodrug in that the IFN-α2b moiety is masked by a PEG group through a protease-cleavable linker rendering the drug inactive in the systemic circulation, thus strongly reducing systemic toxicity. Once the drug accumulates at the tumor site by PD-L1 antibody targeting, the linker is cleaved by proteases that are highly expressed in the tumor environment to achieve specific activation only at the tumor site. This unique property of TJ-L1IF has been confirmed in a series of in vitro and in vivo studies, in which TJ-L1IF demonstrated plasma stability, good safety in cynomolgus monkeys, and superior anti-tumor activity in the PD-1/PD-L1 resistant tumor models, than that achieved by PD-L1 antibody or IFN-α used either alone or in combination. After the first dose of treatment, the active format of the drug was quickly detected and accumulated in the tumor but not in the periphery, confirming the local delivery and conversion to an active form of IFN-α at the tumor site (Figure 2). TJ-L1IF was developed using Affinity's TMEA technology and is now under pre-clinical development.
Figure 2. In vivo anti-tumor activity of TJ-L1IF in PD-L1 resistant tumor model. (a) NSG mice transplanted subcutaneously with colon cancer cell line were treated with Tecentriq (10 mg/kg) alone, Tecentriq (10 mg/kg) and PEG-IFNa (1 mg/kg) combination and TJ-L1IF (3 mg/kg) twice a week. (b) The concentration of PEG cleaved active and PEG masked L1IF was measured in tumor and serum, respectively at 8h and 48h post the first dosing. The ratio of the level of active to that of masked L1IF was calculated.
TJ-C64B is the Company's third bispecific molecule being developed by leveraging a conditional 4-1BB platform which has the advantage of minimizing liver toxicity with an increased therapeutic window. It is specifically designed to simultaneously target Claudin 6 (CLDN6), uniquely expressed in specific cancer types, including ovarian cancer cells, and 4-1BB expressed by T cells to mediate the T cell killing of CLDN6+ tumor cells. CLDN6 is hardly detectable in normal adult tissues to ensure treatment specificity for ovarian cancers. The Company has achieved candidate selection and is actively progressing the pre-clinical development of the candidate molecule.
TJ-C64B activates T cells through 4-1BB stimulation only upon CLDN6 engagement, providing a localized immune activation in tumors with expected efficacy and reduced systemic toxicity. Owing to a competent Fc, TJ-C64B has an added advantage of specifically depleting CLDN6-expressing tumor cells and intra-tumor regulatory T cells highly expressing 4-1BB, which differentiates it from other 4-1BB bispecific antibodies under clinical development. As published in AACR 2022, pre-clinical data showed that TJ-C64B enhances CLDN6-dependent T cell activation upon the engagement of cancer cell lines with different CLDN6 expression levels. In a syngeneic mouse model, TJ-C64B treatment induces strong anti-tumor activity with complete tumor regression in all tested mice at the dose of 4.5 mg/kg and long-term protection from tumor re-challenge through the immunological memory response. Further, ex vivo analysis confirms localized immune activation by TJ-C64B as evident by the increased CD8+ T cells, specifically those residing in tumors (Figure 3). TJ-C64B is now under pre-clinical development and the Company plans to submit an IND in the
Figure 3. In vivo anti-tumor activity of different doses of TJ-C64B treatment. (a) Humanized 4-1BB mice transplanted MC38 tumor cells were treated with different doses of TJ-C64B once a week. After the stop of the treatment, the mice with complete tumor regression were injected with new tumor cells for tumor re-challenge. (b) The percentage of CD8+ T cells in tumor-infiltrating lymphocytes from different treatment groups was analyzed by flow cytometry.
Furthermore, the third wave of innovation initiatives launched in 2021 is making good progress, with multiple innovative molecules now in candidate selection and pre-clinical stage. Among these programs, immune adjuvants are an area of focus designed to prime and amplify both innate and adaptive immune responses. The immune adjuvant portfolio is comprised of cytokine fusions and immuno-cytokines. The cytokine fusions are engineered to extend half-life with Fc fusion and detune the binding affinity and potency of natural cytokine for better safety and efficacy with site-specific mutation. The immuno-cytokines are designed to conjugate engineered cytokines e.g. by masking technology to the tumor or immune cell targeting antibody to achieve pro-longed half-life, minimal systemic toxicity, and enhanced activity by selective tumor targeting.
This growing new portfolio of novel drug candidates represents the Company's strong commitment to sustaining the global competitiveness of its pipeline through continued innovation.
Business Development and Partnership Deals
(1) Research partnerships at
(2) Commercial partnerships are designed as the key model for near-term commercialization of upcoming products, including felzartamab and eftansomatropin alfa. For eftansomatropin alfa, the Company has been working closely with its commercial partner, Jumpcan, to prepare for the product launch and the subsequent local manufacturing plan. The Company is working towards a similar commercial partnership for felzartamab with an aim to maximize the value of felzartamab without investing heavily to build up its own sales capability.
(3) Out-licensing deals continue to remain one of the Company's corporate priorities. The Company is currently pursuing potential global partnership deals of uliledlimab and TJ-CD4B.
Near-Term Product Commercialization
With the rapid progress in clinical development, the Company's most advanced assets, i.e. felzartamab, eftansomatropin alfa, and followed by lemzoparlimab, are expected to potentially achieve BLA in
For felzartamab, there are about 20,000 newly diagnosed MM patients in
For eftansomatropin alfa, approximately 3.4 million children suffer from growth hormone deficiency (PGHD) in
|
[1] |
|
[2] Frost & Sullivan |
Updates Regarding Holding Foreign Companies Accountable Act (HFCAA)
During the last six months, the Company has completed the process of evaluating and selecting a
Environmental, Social and Governance (ESG) Update
In
Currently, females account for two-thirds of the Company's total workforce, and more than 30% of the Company's Board of Directors. The Company has previously established the
In response to urgent situations caused by Covid-19 in 2022,
In the future, the Company intends to continue to strengthen its efforts toward sustainable development and continue to strive to make consequential contributions to society.
Corporate Development
The Company further strengthened its corporate governance and senior management team:
I-Mab appointed Mr.Richard Yeh as Chief Operating Officer and Dr.John Hayslip as Chief Medical Officer onApril 28, 2022 .Mr. Yeh is based inShanghai, China . He was also appointed to joinI-Mab's Board.Mr. Yeh leadsI-Mab's investor relations, global alliance management, and major facilities across the world.Dr. Hayslip is based inthe United States . AsI-Mab's Chief Medical Officer,Dr. Hayslip leads the Company's pipeline development, addressing the key challenges in clinical sciences to increase the probability of success and the speed of clinical development forI-Mab's innovative assets.- Appointment of Dr.
Lin Li , Ph.D. nominated byHony Capital , as a member of the Company's board of directors and a member of the Audit Committee, will become effective onAugust 31, 2022 , replacing Ms.Xi Liu ofHony Capital , who resigned on the same day.Dr. Li served as the Company's director fromJuly 2018 toApril 2020 .Dr. Li has served as a partner sinceMarch 2021 and an investment director fromDecember 2016 toMarch 2021 atHony Capital .Dr. Li worked as an associate atSnow Lake Capital (HK) Limited fromNovember 2014 toNovember 2016 .Dr. Li served as a senior investment manager in the cross-border investment group atHony Capital fromApril 2012 toOctober 2014 . Prior to that, he worked as an associate in the corporate finance department ofGoldman Sachs Gao Hua Securities Company Limited inBeijing fromJuly 2010 toApril 2012 .Dr. Li received his bachelor's degree in biology fromPeking University inJuly 2000 , Ph.D. in biology fromBoston University in 2006, and a Master's degree in business administration fromHarvard Business School in 2010. - Dr. Zheru Zhang resigned from his position as the President of the Company and a director of the board effective from
August 31, 2022 and was appointed as the President for I-Mab Hangzhou, an investee of the Company with a comprehensive biologics manufacturing facility inHangzhou, China . Mr. Jielun Zhu resigned from his position as the Chief Strategy Officer of the Company onJuly 31, 2022 to pursue other interests.
The Company and its senior management demonstrated full confidence in the Company's fundamentals by implementing share purchase plans:
- The Company announced on
August 23, 2022 , that it plans to implement share repurchases pursuant to the share repurchase program previously authorized by its board of directors. On the same day, the Company was informed by Dr.Jingwu Zang , Chairman and Acting Chief Executive Officer of the Company, and other members of senior management of their intention to use personal funds to purchase the Company's American Depositary Shares (the "ADSs") on the open market. Under the share purchase plans, the Company and the senior management may purchase up toUS$40 million of ADSs in aggregate. The timing and dollar amount of share repurchase and share purchase transactions will be subject to the applicableU.S. Securities and Exchange Commission rule requirements. The Company's board of directors will review the implementation of share repurchases periodically and may authorize adjustment of its terms and size. - In
January 2022 , the Company's senior management executed a share purchase plan and purchased over 78,000 ADSs in aggregate on the open market.
The Company invested in I-Mab Hangzhou in 2020 as a part of the Company's overall strategic plan. On
The Company was ranked among the top companies in six different categories by the leading global financial publication Institutional Investor, based on its 2022 All-Asia Executive Team survey. The awards recognize
The Company recently hosted an R&D Day, providing a comprehensive update on its business strategy, clinical development of its key innovative assets as well as its next-generation preclinical programs, and the Company shared key repositioning strategies.
First-Half 2022 Financial Results
Cash Position
As of
Net Revenues
Total net revenues for the six months ended
Research & Development Expenses
Research and development expenses for the six months ended
Administrative Expenses
Administrative expenses for the six months ended
Other Income (Expenses), net
Net other expenses for the six months ended
Equity in loss of affiliates
Equity in loss of affiliates for the six months ended
Net Loss
Net loss for the six months ended
Non-GAAP Net Loss
Non-GAAP adjusted net loss, which excludes share-based compensation expenses, for the six months ended
Subsequent Event
As a subsequent event, on
Conference Call and Webcast Information
The Company's management will host conference calls to discuss the results and updates, and a Mandarin session conference call will be held at
|
Meeting URL: |
https://i-mabbiopharma.zoom.us/j/81457574870?pwd=ZTJ0SVIzMWpJS2Q0WlJiTGROL1Bndz09 |
|
Meeting ID: |
814 5757 4870 |
|
Password: |
593909 |
English Session
|
Meeting URL: |
https://i-mabbiopharma.zoom.us/j/84196628861?pwd=K3hpeEpMUUdXUGFlWmU4dlF3UWtnZz09 |
|
Meeting ID: |
841 9662 8861 |
|
Password: |
934660 |
About
I-Mab Forward Looking Statements
This announcement contains forward-looking statements. These statements are made under the "safe harbor" provisions of the
Use of Non-GAAP Financial Measures
To supplement its consolidated financial statements which are presented in accordance with
Non-GAAP information is not prepared in accordance with GAAP and may be different from non-GAAP methods of accounting and reporting used by other companies. The presentation of this additional information should not be considered a substitute for GAAP results. A limitation of using adjusted net income (loss) is that adjusted net income (loss) excludes share-based compensation expense that has been and may continue to be incurred in the future.
Exchange Rate Information
This announcement contains translations of certain RMB amounts into
|
|
||||
|
Consolidated Balance Sheets |
||||
|
(All amounts in thousands, except for share and per share data, unless otherwise noted) |
||||
|
As of |
As of |
|||
|
2021 |
2022 |
|||
|
RMB |
RMB |
US$ |
||
|
Assets |
||||
|
Current assets |
||||
|
Cash and cash equivalents |
3,523,632 |
3,710,901 |
554,023 |
|
|
Accounts receivable |
33,081 |
510 |
76 |
|
|
Contract assets |
253,780 |
291,079 |
43,457 |
|
|
Short-term investments |
753,164 |
211,184 |
31,529 |
|
|
Inventories |
27,237 |
- |
- |
|
|
Prepayments and other receivables |
190,824 |
101,004 |
15,080 |
|
|
Total current assets |
4,781,718 |
4,314,678 |
644,165 |
|
|
Property, equipment and software |
45,716 |
61,141 |
9,128 |
|
|
Operating lease right-of-use assets |
112,781 |
100,860 |
15,058 |
|
|
Intangible assets |
119,666 |
119,277 |
17,808 |
|
|
|
162,574 |
162,574 |
24,272 |
|
|
Investments accounted for using the equity method |
352,106 |
217,662 |
32,496 |
|
|
Other non-current assets |
26,634 |
15,380 |
2,296 |
|
|
Total assets |
5,601,195 |
4,991,572 |
745,223 |
|
|
Liabilities and shareholders' equity |
||||
|
Current liabilities |
||||
|
Accruals and other payables |
593,335 |
547,472 |
81,736 |
|
|
Operating lease liabilities, current |
30,669 |
42,527 |
6,349 |
|
|
Total current liabilities |
624,004 |
589,999 |
88,085 |
|
|
Put right liabilities |
96,911 |
70,242 |
10,487 |
|
|
Contract liabilities |
224,000 |
240,006 |
35,832 |
|
|
Operating lease liabilities, non-current |
81,786 |
61,302 |
9,152 |
|
|
Other non-current liabilities |
14,934 |
13,948 |
2,082 |
|
|
Total liabilities |
1,041,635 |
975,497 |
145,638 |
|
|
Shareholders' equity |
||||
|
Ordinary shares ( |
126 |
131 |
20 |
|
|
Additional paid-in capital |
9,100,777 |
9,370,583 |
1,398,991 |
|
|
Accumulated other comprehensive income (loss) |
(186,510) |
47,051 |
7,025 |
|
|
Accumulated deficit |
(4,354,833) |
(5,401,690) |
(806,451) |
|
|
Total shareholders' equity |
4,559,560 |
4,016,075 |
599,585 |
|
|
Total liabilities and shareholders' equity |
5,601,195 |
4,991,572 |
745,223 |
|
|
|
|||||
|
Consolidated Statements of Comprehensive Loss |
|||||
|
(All amounts in thousands, except for share and per share data, unless otherwise noted) |
|||||
|
For the six months ended |
|||||
|
2021 |
2022 |
||||
|
RMB |
RMB |
US$ |
|||
|
Revenues |
|||||
|
Licensing and collaboration revenue |
17,775 |
23,756 |
3,547 |
||
|
Supply of investigational products |
- |
28,102 |
4,195 |
||
|
Total revenues |
17,775 |
51,858 |
7,742 |
||
|
Cost of revenues |
- |
(27,237) |
(4,066) |
||
|
Expenses |
|||||
|
Research and development expenses (Note 1) |
(592,993) |
(452,618) |
(67,574) |
||
|
Administrative expenses (Note 2) |
(451,500) |
(392,460) |
(58,593) |
||
|
Loss from operations |
(1,026,718) |
(820,457) |
(122,491) |
||
|
Interest income |
9,409 |
6,566 |
980 |
||
|
Other income (expenses), net |
51,904 |
(51,944) |
(7,755) |
||
|
Equity in loss of affiliates (Note 3) |
(114,200) |
(181,022) |
(27,026) |
||
|
Loss before income tax expense |
(1,079,605) |
(1,046,857) |
(156,292) |
||
|
Income tax benefit |
3,124 |
- |
- |
||
|
Net loss attributable to |
(1,076,481) |
(1,046,857) |
(156,292) |
||
|
Net loss attributable to ordinary shareholders |
(1,076,481) |
(1,046,857) |
(156,292) |
||
|
Net loss attributable to |
(1,076,481) |
(1,046,857) |
(156,292) |
||
|
Foreign currency translation adjustments, net of nil tax |
(73,577) |
233,561 |
34,870 |
||
|
Total comprehensive loss attributable to |
(1,150,058) |
(813,296) |
(121,422) |
||
|
Net loss attributable to ordinary shareholders |
(1,076,481) |
(1,046,857) |
(156,292) |
||
|
Weighted-average number of ordinary shares used in calculating net loss per |
168,827,190 |
188,857,353 |
188,857,353 |
||
|
Weighted-average number of ordinary shares used in calculating net loss per |
168,827,190 |
188,857,353 |
188,857,353 |
||
|
Net loss per share attributable to ordinary shareholders |
|||||
|
—Basic |
(6.38) |
(5.54) |
(0.83) |
||
|
—Diluted |
(6.38) |
(5.54) |
(0.83) |
||
|
Net loss per ADS attributable to ordinary shareholders (Note 4) |
|||||
|
—Basic |
(14.67) |
(12.74) |
(1.90) |
||
|
—Diluted |
(14.67) |
(12.74) |
(1.90) |
||
|
Note: |
|||||
|
(1) Includes share-based compensation expense of |
|||||
|
(2) Includes share-based compensation expense of |
|||||
|
(3) Includes share-based compensation expense of |
|||||
|
(4) Each ten ADSs represents twenty-three ordinary shares. |
|||||
|
|
||||||
|
Reconciliation of GAAP and Non-GAAP Results |
||||||
|
For the six months ended |
||||||
|
2021 |
2022 |
|||||
|
RMB |
RMB |
US$ |
||||
|
GAAP net loss attributable to |
(1,076,481) |
(1,046,857) |
(156,292) |
|||
|
Add back: |
||||||
|
Share-based compensation expense |
347,061 |
198,867 |
29,690 |
|||
|
Non-GAAP adjusted net loss attributable to |
(729,420) |
(847,990) |
(126,602) |
|||
|
Non-GAAP adjusted loss attributable to ordinary |
(729,420) |
(847,990) |
(126,602) |
|||
|
Weighted-average number of ordinary shares used in |
168,827,190 |
188,857,353 |
188,857,353 |
|||
|
Weighted-average number of ordinary shares used in |
168,827,190 |
188,857,353 |
188,857,353 |
|||
|
Non-GAAP adjusted loss per share attributable to |
||||||
|
—Basic |
(4.32) |
(4.49) |
(0.67) |
|||
|
—Diluted |
(4.32) |
(4.49) |
(0.67) |
|||
|
Non-GAAP adjusted loss per ADS attributable to ordinary |
||||||
|
—Basic |
(9.94) |
(10.33) |
(1.54) |
|||
|
—Diluted |
(9.94) |
(10.33) |
(1.54) |
|||
![]() View original content to download multimedia:https://www.prnewswire.com/news-releases/i-mab-provides-business-and-corporate-updates-and-reports-financial-results-for-the-six-months-ended-june-30-2022-301614531.html
View original content to download multimedia:https://www.prnewswire.com/news-releases/i-mab-provides-business-and-corporate-updates-and-reports-financial-results-for-the-six-months-ended-june-30-2022-301614531.html
SOURCE